A new catalyst that converts cellulose into high yields of ethylene glycol could help reduce our dependence on fossil fuels in making plastics, say US-based scientists.
Ethylene glycol (EG) is an important petroleum product used to make polyester fibres and resins for the plastics industry, as well as antifreeze. Global demand exceeds 17 million tonnes per year. It is already possible to produce the chemical from cellulose - and therefore use waste materials from the forestry and food industries - but only with precious metals such as platinum. Now a team of researchers led by Jingguang Chen at the University of Delaware say their tungsten carbide catalyst not only does the same job for a third of the price, it does it better.
'We were pleasantly surprised that when we used carbide catalysts, we were getting to ethylene glycol directly instead of C6 sugars [such as mannitol and sorbitol],' says Chen. The platinum process, he explains, turns cellulose into six-carbon sugars, but has to be followed by other conversions to get to feedstock chemicals like the two-carbon ethylene glycol.
The direct conversion, which uses water, cellulose, and tungsten carbide (W2C) deposited on a carbon support, is carried out at 245 degrees Celsius and under a 60 atmosphere pressure of hydrogen gas. The researchers found that by adding a small amount of nickel to the reaction they could produce more EG, increasing the yield from 29 per cent to 61 per cent - the highest yet achieved. But they are still trying to understand how the catalyst works on a molecular level.
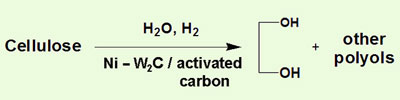
High-pressure nickel/tungsten carbide chemistry converts cellulose directly to ethylene glycol, an intermediate chemical for the plastics industry
|
Chen says several patents have been filed for the method, but he admits it would require substantial investment for scale-up to industry levels. 'This is relatively high pressure chemistry, so the reactor design is not straightforward. Industry has to be interested in it and start putting in capital investment.'
The team will try to optimise the reaction under more favourable conditions. But Derek Atkinson, Business Development Director for Oxford Catalysts, says that to appeal to industry the process would have to outdo not just other cellulose to EG processes, but the conventional petroleum to EG process. He also thinks industry is more keenly focused on turning biomass into fuel than into chemicals.
'When we think of biomass, we think of gasifying it and producing biofuels,' says Atkinson. 'The economics for biomass to fuels can be a little woollier, because there's a government mandate for a certain proportion of the fuels to come from a biosource, whereas for a process from biomass to ethylene glycol you'd have to come up with a process that's inherently cheaper than the current process.'
Hayley Birch
References




