An important insight into the mechanism of hydrocarbon reactions has been revealed by chemists working in France.
Catalysed transformations of hydrocarbons are important in the petrochemical industry but their inertness must be overcome by working at high temperature and pressure. Jean Sommer and colleagues at the University of Strasbourg have shown the presence of carbenium ions in hydrocarbon transformation on zeolite mineral catalysts. These ions are generally accepted as reaction intermediates but their formation is still not fully understood.
Sommer used a combination of NMR measurements and labelled starting materials to detect the presence of the carbenium ions. He explained: 'We show the existence of these intermediates on zeolites even at room temperature, a controversial point in solid acid catalysis.'
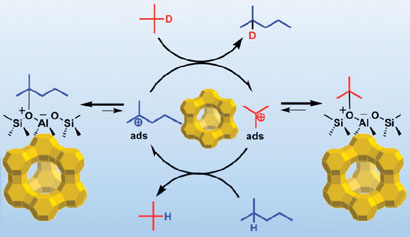
Carbenium ions intermediates on zeolites have been seen using NMR
|
'Carbenium ions are generally present at very low concentration, and thus difficult to detect and observe,' Sommer continued. 'Our work confirms that they are necessary intermediates to make sense of hydrocarbon reactivity.'
Pierre Esteves, an expert in carboacation chemistry at the Federal University of Rio de Janeiro, Brazil, comments, 'the elegant use of NMR coupled with isotopically labelled hydrocarbons shows that strong evidence for carbocations behaviour within these porous catalysts, even at room temperature.'
The next step, according to Sommer, is to find out if the reactions take place inside the zeolite cavities or on the outer surface of the catalyst.
Michael Spencelayh
Enjoy this story? Spread the word using the 'tools' menu on the left or add a comment to the Chemistry World blog.




