Real-time, atomic level microscopy has revealed that the round, cage-like structures of fullerenes can form directly from sheets containing large numbers of carbon atoms. The resulting 'top down' model diverges from a large body of evidence supporting bottom up mechanisms in which near atomic carbon is needed to produce fullerenes.
Fullerenes have presented scientists with a puzzle ever since their discovery 25 years ago: how can a single, flat layer of carbon atoms arranged in a hexagonal pattern (graphene) transform into a ball containing only pentagons? The answer has nearly always involved breaking down the sheet into single or paired carbon atoms.
Now, a group led by Andrei Khlobystov at the University of Nottingham, UK, has found a way to watch the process as it happens - using a low-voltage transmission electron microscopy (TEM) methodology developed by team member Andrey Chuvilin. 'It's a fantastic methodology, which allows you literally to film chemical reactions at atomic level,' says Khlobystov. 'So that's the chemist's dream come true.'
Combining their footage with quantum chemical models - to prove that the process made sense thermodynamically - the team outlined a process for fullerene formation that Khlobystov thinks is simpler than previously proposed bottom up mechanisms. It is driven by the fact that all carbon atoms 'want' to have three bonds. So when one atom is removed at the edge of the sheet, leaving the two adjacent carbons with fewer than the desired three bonds, they immediately join together to create a pentagon and the sheet begins to curl up. As more atoms are chipped away via electron beam, a bowl-like structure forms, which finally zips up to form a ball.
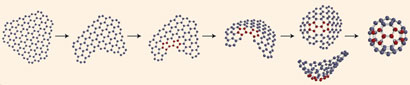
The graphene sheet curls up, then forms a bowl-like structure, and finally zips up into a ball
© Nature Chemistry
|
As Khlobystov explains, the size of the graphene flake is critical - the 'sweet spot' is between 60-100 carbon atoms. If the flake is too large, the initial curvature makes little difference to the flake as a whole, whereas if it is too small, the bonds will be strained.
The process Khlobystov's team has unveiled is beautiful, says Boris Yakobson, a nanocarbon expert at Rice University in Texas, US. 'The authors have caught it in action, and also offer a logical explanation of why, possibly, this happens,' he says. But he doubts the same mechanism is central to real fullerene production.
Khlobystov, however, believes an analogy can be drawn. 'I know that conditions in the TEM are quite unusual, but imagine that the electron beam you pass through the sample plays the role of high temperature - it helps to remove some carbon atoms at the edge. The high temperature in [fullerene production] will do the same thing.'
Hayley Birch
Interesting story? Spread the word using the 'tools' menu on the left.




