Abstract
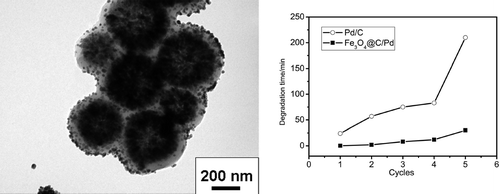
Fe3O4 microsphere is a good candidate as support for catalyst because of its unique magnetic property and large surface area. Coating Fe3O4 microspheres with other materials can protect them from being dissolved in acid solution or add functional groups on their surface to adsorb catalyst. In this paper, a carbon layer was coated onto Fe3O4 microspheres by hydrothermal treatment using polyethylene glycol as the connecting agents between glucose and Fe3O4 spheres. Through tuning the added amounts of reactants, the thickness of the carbon layer could be well-controlled. Because of the abundant reductive groups on the surface of carbon layer, noble metal ions could be easily adsorbed and in situ reduced to nanoparticles (6−12 nm). The prepared catalyst not only had unique antiacid and magnetic properties, but also exhibited a higher catalytic activity toward the reduction of methyl orange than commercially used Pd/C catalyst.
/files/info/2011/am101077a.pdf
