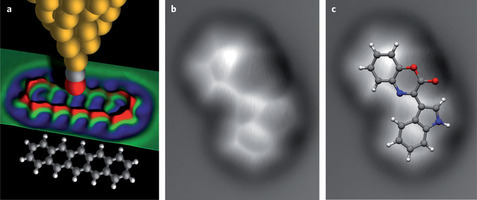
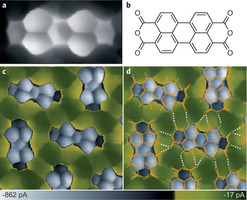
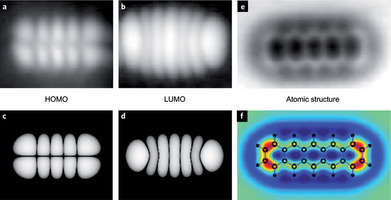
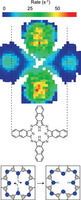
Nature Chemistry | Perspective
Recent advances in submolecular resolution with scanning probe microscopy
- Journal name:
- Nature Chemistry
- Volume:
- 3,
- Pages:
- 273–278
- Year published:
- (2011)
- DOI:
- doi:10.1038/nchem.1008
- Published online
- Лаборатория оксидных систем
- Лаборатория нестехиометрических соединений
- Лаборатория гетерогенных процессов
- Лаборатория неорганического синтеза
- Лаборатория химии соединений редкоземельных элементов
- Лаборатория структурного и фазового анализа
- Лаборатория физико-химических методов анализа
- Лаборатория квантовой химии и спектроскопии им. профессора А.Л. Ивановского
- Лаборатория физико-химии дисперсных систем
- Лаборатория перспективных функциональных материалов для химических источников тока
- Лаборатория ионики твёрдого тела
- Группа патентно-конъюнктурных исследований